 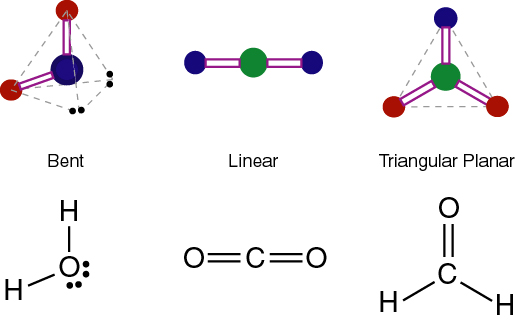
The covalent bonds can be polar or nonpolar depending upon the various factors. The polarity of a molecule is decided by the type of bonding it involves across its atoms. The dipole across the C=O bonds is non zero and originates towards oxygen, but the equal and opposite dipoles cancel out each other resulting in a nonpolar molecule. And oxygen being more electronegative pulls the bonded electron pair slightly more towards its side and gains partial negative charge and carbon gains partial positive charge. The oxygen atoms lie at both sides of the carbon atom. Oxygen needs 2 electrons to complete its octet and carbon needs 4 for the same.Īs a result, two oxygen atoms and carbon atom share two electrons of each other forming double bonds across carbon and oxygen atoms and fills their octet to get stabilized. The valence electrons of oxygen are 6 and that of carbon is 4. The symmetrical geometry (linear) of the CO2 molecule cancels out the polarity of the C=O polar bonds due to which net dipole of the molecule becomes zero. The molecule of CO2 is polar because of the following reason. The CO2 molecule is nonpolar and its dipole also turns out to be 0 D.Ĭonclusion Why is CO2 a Nonpolar Molecule? The difference between the electronegativity of both atoms is sufficient to have polarity across C=O bonds in the CO2 molecule.īut due to the symmetry of the molecule ie linear structure, both C=O bonds lie opposite to each other, and polarity of both gets canceled by each other. The electronegativity of carbon is 2.55 and that of oxygen is 3.44. The shape of the molecule is linear as both oxygen atoms are opposite to each other. 
mol−1Ĭarbon is the central atom surrounded by the two oxygen atoms on both sides.Mol mass of CO2 = 1 * 12(Mol mass of C) 2 * 16 (Mol mass of O) = 44.009 g If we talk about the chemical composition of the CO2 molecule, it consists of one carbo atoms and two oxygen atoms. Rivers, lakes, etc.Īpart from humans, it is released by various aerobic organisms on earth were they metabolize the carbohydrates to release carbon dioxide gas. It is soluble in water therefore also occurs in various water bodies like groundwater. Its naturally occurring sources are like humans, geysers, volcanoes, and many chemical reactions including the dissolution of carbonate in water and acid. It is a naturally occurring gas in the earth’s atmosphere. Although the carbon and oxygen differ in their electronegativity due to which C=O bond is polar, the polarity of both opposite C=O bonds get canceled by each other due to symmetrical shape and result in a nonpolar molecule with zero dipole moment.Ĭarbon dioxide gas is colorless and odorless in nature and its density is 60% higher than that of dry air around us. So, Is CO2 polar or nonpolar? CO2 (carbon dioxide) is a nonpolar molecule because of its linear symmetric shape. We will discuss this and will cover its properties and uses. Many of you may have doubts regarding whether the CO2 gas molecule is polar or not. We, humans, are also the source of this gas as we release carbon dioxide through our body in different forms. \) Some examples of polar molecules based on molecular geometry (HCl, NH3 and CH3Cl).Carbon dioxide is a colorless gas with chemical formula CO2 present in our environment with no odor at low concentration. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
